Patient Monitoring Arm Band
MDR Class IIa
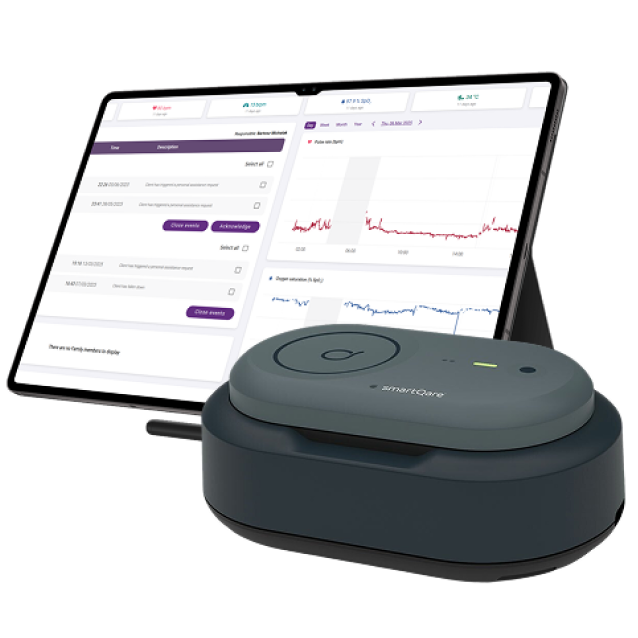
The goal of this arm band is to enhance patient quality of life, reduce medical costs, support healthcare professionals in managing larger patient groups, and enable faster responses to medical concerns.
Analysis
Connectivity
Features
Certificates and device classes

Contribution
Thaumatec was responsible for the full firmware development, including architecture design, technology selection, software implementation, testing, quality assurance, and CI/CD infrastructure setup. Additionally, our team contributed to biomedical algorithm research, cloud device protocol specifications, and integration activities.
Solution
The solution consists of a wearable device, an online platform, and a mobile app. The arm band features multiple sensors, including PPG, accelerometer, temperature, and pressure sensors, to measure vital biomedical parameters and generate alerts in emergency situations. Continuous cloud communication provides real-time feedback to users. The development process adhered to ISO 13485 certification standards.





